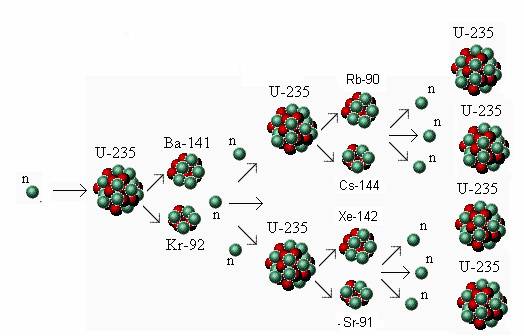 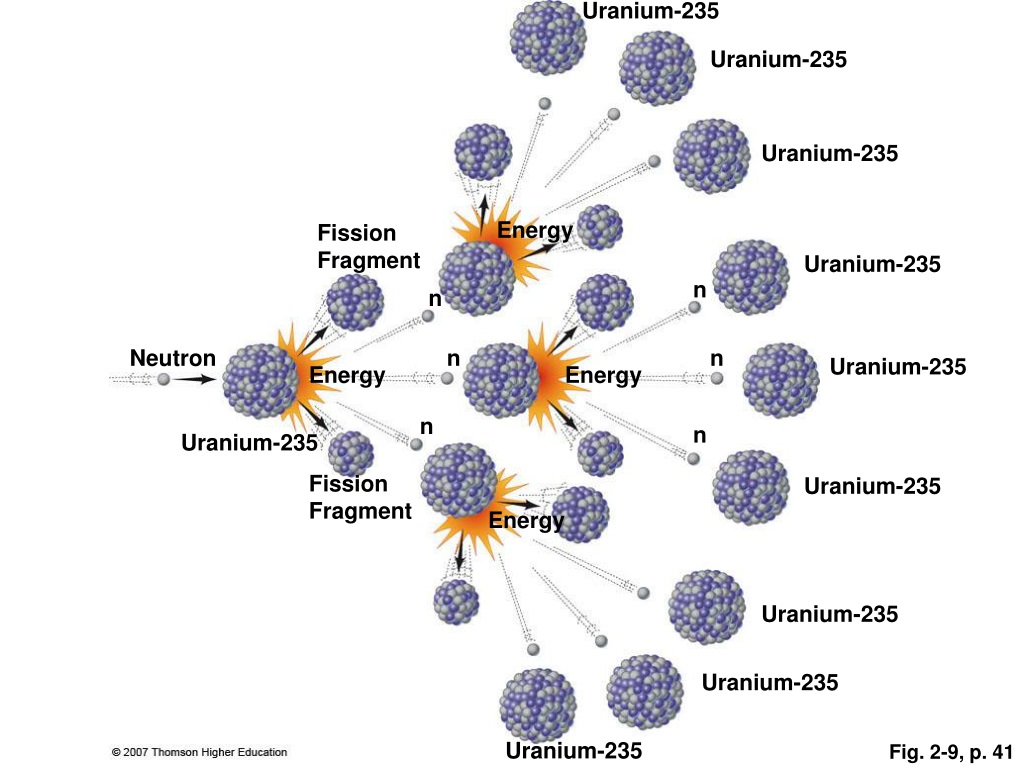
Pathway one: Provide a source of sufficiently energetic neutrons to induce fissions without a chain reaction. Two alternative physics pathways exist to make use of the two naturally abundant actinide isotopes, uranium-238 and thorium-232, as follows: Indeed, the total utilization of mined uranium is only about 1%, with 99% discarded as depleted uranium or as the main component of spent nuclear fuel (SNF). Light water reactors (LWRs), the reactor design currently responsible for producing the majority of the world's nuclear power, rely on the rare uranium-235 isotope as fuel, leaving most of the uranium-238 isotope unused along with the thorium-232 isotope which LWRs entirely ignore. Isotopic enrichment is a difficult industrial process in which a mixture of two or more isotopes of an element is divided into two different mixtures, an “enriched” mixture with an increased concentration of one isotope and a “depleted” mixture with a depressed concentration of the same isotope. A sustained fission chain reaction is impossible with either of these more plentiful isotopes. However, they are only fissionable, not fissile. Both can be fissioned, releasing about 200 MeV of energy per atom. Uranium-238, which has a 4.47 billion year half-life is 138 times more abundant and thorium-232, with a 14.05 billion year half-life, is about 500 times more abundant. Uranium-235 with a 704 million year half-life is the only naturally occurring fissile isotope. Only the fissile actinide isotopes can support fission chain reactions, since emitted fission daughter neutrons having enough energy to fission other non-fissile actinides are rare. Examples of fissile actinide isotopes include uranium-233, uranium-235, plutonium-239 and plutonium-241, but of these only uranium-235 is found in nature. Actinide isotopes can be classified according to whether they are fissile, meaning that they can be fissioned by slow neutrons having room-temperature thermal motion energies of about 0.025 eV. Instability and ease of fission of at least some actinide isotopes generally increases with ascending atomic number and spontaneous fission also becomes common in the higher actinides.Īll actinide atoms are fissionable, meaning each can be fissioned if its atomic nucleus is struck by a sufficiently energetic neutron. All actinide isotopes are unstable to radioactive decay involving emission of alpha or beta particles along with gamma rays, as also are all isotopes of the next five atomic numbers below the actinides. There are 15 actinide elements, each with several important isotopes. The entire teachings of the above applications are incorporated herein by reference. This application claims the benefit of U.S.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
